Video
1 / 6
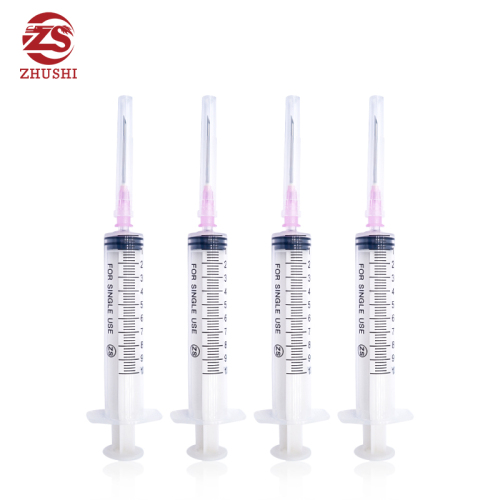
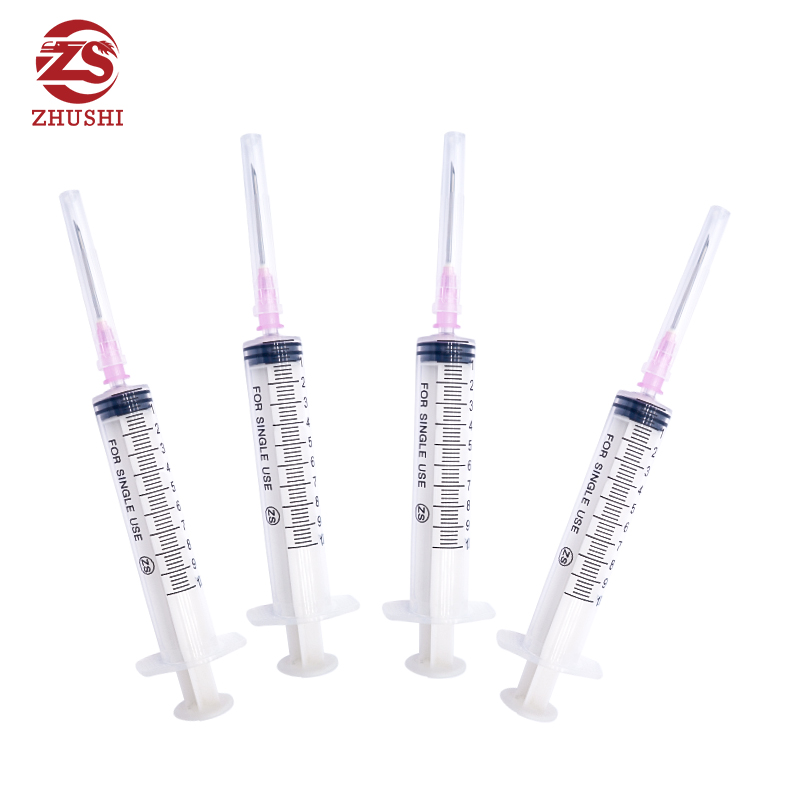
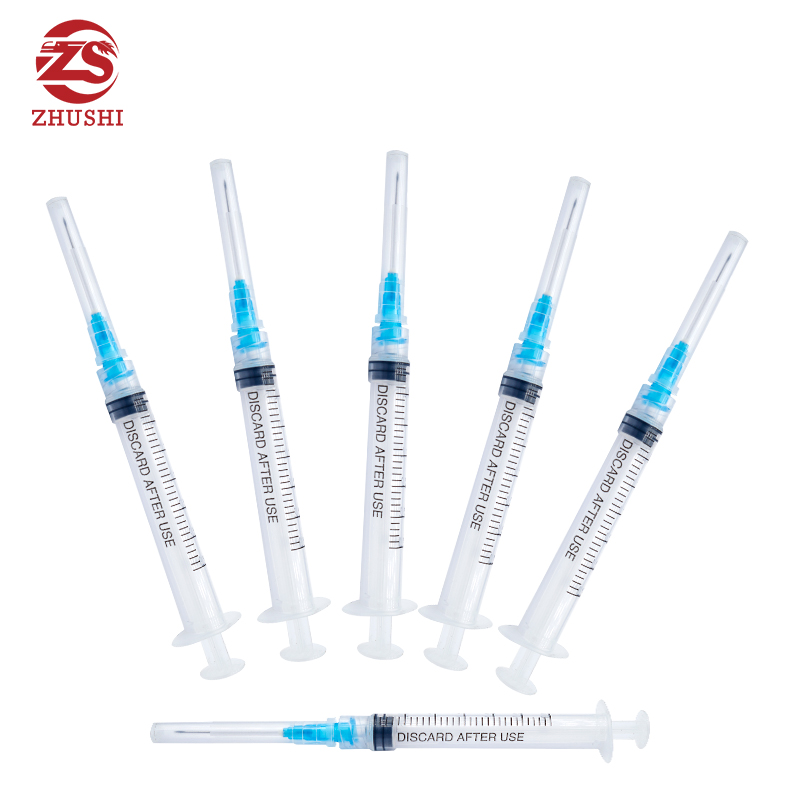
High quality disposable medical syringe
Options:
- 10ml
| Brand Name : | ZS |
|---|---|
| place of origin : | China |
Heze, Shandong, China
- Manufacturer
- OEM service
- Gold Supplier

- Platform Certification

- SGS Certification

Product description

High quality disposable medical syringe
1) Disposable syringe with three parts,luer lock or luer slip
2) Passed CE and FDA certification.
3) Transparent barrel allows easy measurement of the volume contained in the syringe.
4) Graduation printed by indeliable ink on the barrel is easy to read
5) The plunger fits the inside of barrel very well to allow for smooth movement
6) Material of barrel and plunger:Material grade PP(Polypropylene)
7) Materials of gasket: Synthetic Rubber(latex free)
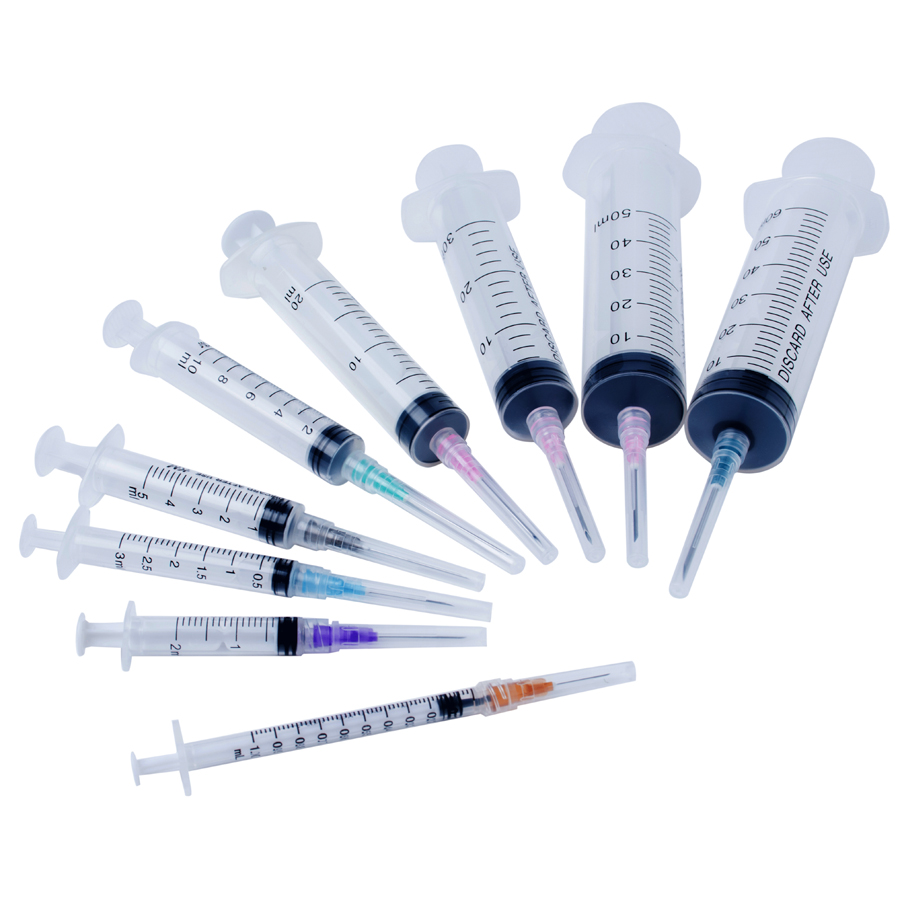
8) Products 1ml,3ml,5ml,10ml with Blister packing are available.
9) Sterilized by EO gas, non-toxic and non-pyrogenic.
Shelf life: 3 years
Steril insulin syringe is available too.

Item
10ml syringe
Material
pp
Standard
IS013485
Features
Safety well and close it,comfort,convenience and economy
Sample
Free of charge
Product Certifications
CE
Factory Certifications
IS09001,ISO14001
1 ml l 3ml ml 5ml Syringe disposable sterile insulin syringe with needle
The company profile:
Our company has been established for 20 years. We own three GMP workshops which can produce all kinds of syringes and infusion sets and so on.
Disposable syringes:
three parts include tube,plunger and piston.
luer slip or luer lock
with needle or without needle
the latex piston or the latex free piston
the PE or Blister individual package
the PE or Box secondly package

Shandong Zhushi Pharmaceutical Group Co., Ltd. was established in 2003, located in Shanxian County, Heze city, Shandong province, China. With a registered capital of 100 million yuan, more than 5000 employees, our company has several factories including production,research and development,we produce medical consumables ,epidemic prevention products,medical gel and patches, skin care and cosmetics.Our company covers an area about 380,000m², our annual income is 3 billion yuan.The company adopted strict standards to create perfect products and has passed EN13485 certification issued by the British authority.
We establish the quality management system according to ISO9001 and ISO13485 standards, strictly regulate the production
process, use CP, MSA, 5S and other management concepts to strengthen the control of product quality.We have import and export license, electronic port , inspection and quarantine application procedures for companies in accordance with government regulations.After more than ten years of rapid development, the company has formed a collection of production and sales of medical devices, disinfection and sanitary products, polymers, cosmetics, health foods, research and development of biological reagents.We incorporate consulting, training, transportation and printing together. A diversified modern Chinese medicine industry cluster with a core of the pharmaceutical industry.
We are sincerely looking forward to cooperating with more overseas customer based on mutual benefits and long term cooperation.




1,National Medical Products Whitelist
2,We have strict quality control system, Good reputation in the market.
3,All of our products have international certification: ISO,CE and so on.
4,More than 20 years successful OEM/ODM experience for brand product.
5,We have professional R&D team, design team, sale team and production workers.
6,All raw material we used are environmental-friendly;
7,Quality Control Department specially responsible for quality checking in each process.



1.Are you a factory or a trading company?
Re: Factory, more than 20 years experience in Medical Devices held.
2.What services can you offer?
Re: No matter you are smaller buyer or bulk purchaser, you will get quality products and the most professional solution and excellent service from us. we can do FCA, FOB, and CIF.
3.Can we trust you?
Re: Sure, we have CE etc. certificate. we supply for chinese local government, and some oversea goverment department.
4.Can we put our logo or designs on your package?
Re: Yes.we can.OEM ODM service are welcome.


Video
Heze, Shandong, China
- Manufacturer
- OEM service
- Gold Supplier

- Platform Certification

- SGS Certification
Send your inquiry to this supplier