Video
1 / 4
DOA Rapid Test Kit of MOP/MET/COC/THC
Options:
- 50T
- 25T
- 20T
- 15T
- 10T
- OEM
| Model No. : | THC002 |
|---|---|
| Brand Name : | MR |
| Brand Name : | MR |
Changchun, Jilin, China
- Manufacturer
- Trade Company
- OEM service
- Gold Supplier

- Platform Certification


Product description
Test procedure
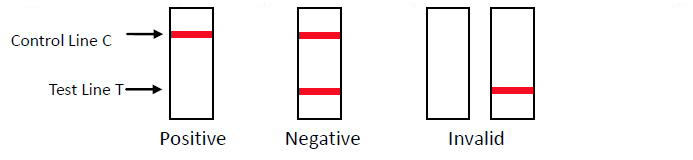
Result Judgment
POSITIVE: One red line appears in the control region(C). No apparent red or pink line appears in the test region (T). NEGATIVE: Two distinct red lines appear. One line should be in the control region (C) and the other line should be in the test region (T). INVALID: No red bands appear or control line fails to appear, indicating that the operator error or reagent failure. Verify the test procedure and repeat the test with a new testing device.
Our main products cover Medical Consumables, Rapid Test Kit, Urinalysis Reagent Strip, Biochemistry Analyzer, Hematology Analyzer, POCT Analyzer,
Products Pictures














Video
Changchun, Jilin, China
- Manufacturer
- Trade Company
- OEM service
- Gold Supplier

- Platform Certification

Send your inquiry to this supplier